
Delayed second dose versus standard regimen for COVID-19 vaccination. cdc.gov/vaccines/covid-19/info-by-product/clinical-considerations.html Interim clinical considerations for the use of mRNA COVID-19 vaccines currently authorized in the United States.cdc.gov/coronavirus/2019-ncov/vaccines/faq.html Frequently asked questions about COVID-19 vaccination.FDA statement on following the authorized dosing schedules for COVID-19 vaccines.fda.gov/news-events/press-announcements/fda-issues-emergency-use-authorization-third-covid-19-vaccine FDA issues emergency use authorization for third COVID-19 vaccine.diseases-conditions/covid-19-vaccine-faq COVID-19 vaccine questions and answers (2021).Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine: supplementary appendix. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. uk/government/uploads/system/uploads/attachment_data/file/949505/annex-a-phe-report-to-jcvi-on-estimated-efficacy-of-single-vaccine-dose.pdf Annex A: Report to JCVI on estimated efficacy of a single dose of Pfizer BioNTech (BNT162b2 mRNA) vaccine and of a single dose of ChAdOx1 vaccine (AZD1222).Early rate reductions of SARS-CoV-2 infection and COVID-19 in BNT162b2 vaccine recipients. You can learn more about how we ensure our content is accurate and current by reading our editorial policy. Healthline has strict sourcing guidelines and relies on peer-reviewed studies, academic research institutions, and medical associations. Researchers will continue to investigate the impact that emerging strains have on current and future COVID-19 vaccines. It’s important to note that this is still a developing area of study. variant were neutralized, researchers saw a 6.4-fold drop in neutralization for the South African strain. 
When compared to an early strain of the novel coronavirus, antibody neutralization of this virus was two-thirds weaker.Ī similar report looked at the ability of antibodies generated by the Moderna vaccine to neutralize test viruses. One report tested antibodies generated by the Pfizer-BioNTech vaccine against a test virus containing the spike protein mutations found in the South African variant. 
Initial data indicates that these vaccines may be less effective against the South African strain. Laboratory (test tube) studies have been performed with these strains and the Pfizer-BioNTech and Moderna vaccines. Two new strains you may have heard a lot about are: There have been concerns about about how effective the vaccines are against these new variants. Over the past several months, new strains, or variants, of SARS-CoV-2 have been identified. At this point, the vaccine was found to be 94.1 percent effective at preventing COVID-19.Ĭan the vaccine protect you against other strains of the new coronavirus? The Moderna clinical trial looked at vaccine effectiveness 2 weeks after participants had received their second dose. Researchers found that the vaccine was 95 percent effective at preventing COVID-19 at this point. The Pfizer-BioNTech clinical trial evaluated vaccine effectiveness 1 week after participants had gotten their second dose. Now that we’ve discussed how long it generally takes to have immunity, let’s take a look at the effectiveness of the Pfizer-BioNTech and Moderna vaccines in the weeks after the second dose. Because of this, you can still become ill during this time frame. In fact, it typically takes about 2 weeks for your body to build up immunity. However, immunity doesn’t happen immediately after vaccination. It can then draw upon this information to protect you if you’re exposed to the actual virus in the future. 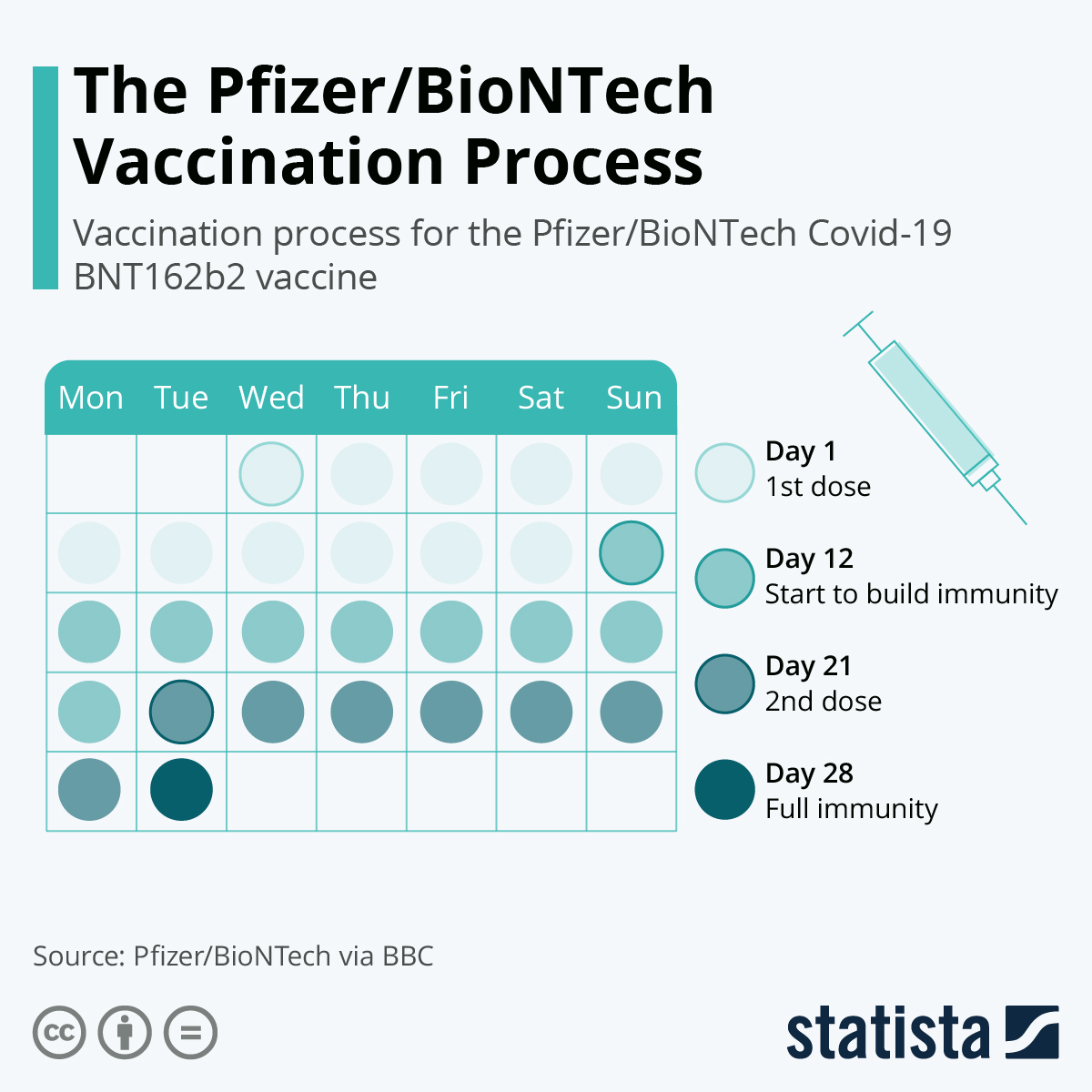
It’s used to help the virus bind to and enter host cells in your body.īecause your immune system has a memory, it can use the vaccine to analyze and store information about the spike protein. This protein is found on the viral surface. How long does it take to have immunity after the second vaccine dose?īoth the Pfizer-BioNTech and Moderna vaccines work by introducing your immune system to a part of the new coronavirus called the spike protein.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
